Our Focus
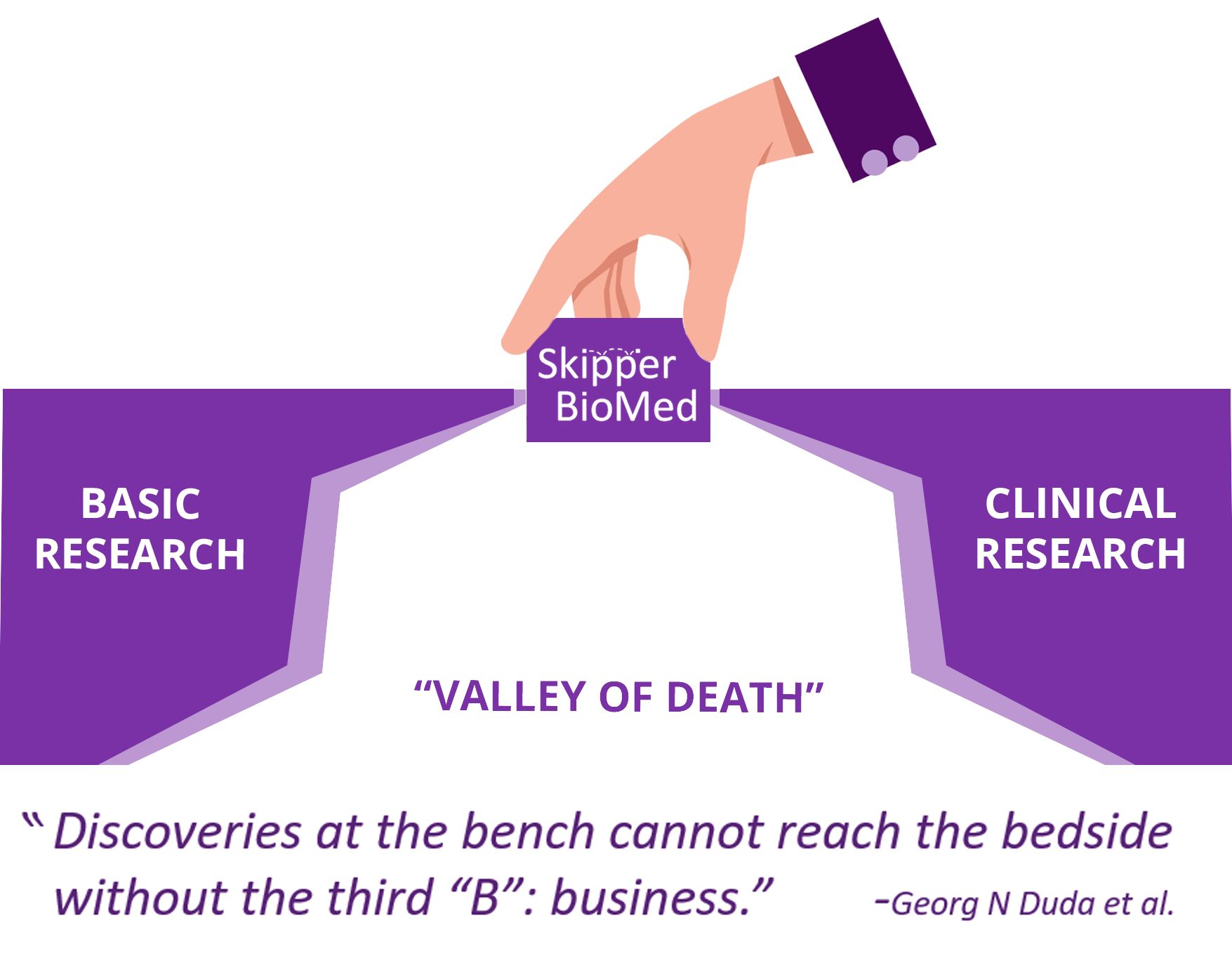
How many times have you seen a news story that touts “new research out of University X could be the cure for cancer”, only to never hear about it again? Not only is this a failure of how science is communicated, interpreted, and distributed to the public, but it also reflects how difficult it is to be able to successfully provide new diagnostics and therapeutics to patients. The process of “translational” research and product development is often depicted as linear, but in reality it is a complicated balancing act of trying to determine what and how much data to generate, which stakeholders/investors should be involved and when, how your product will influence clinical decision making, and why people should believe in you.
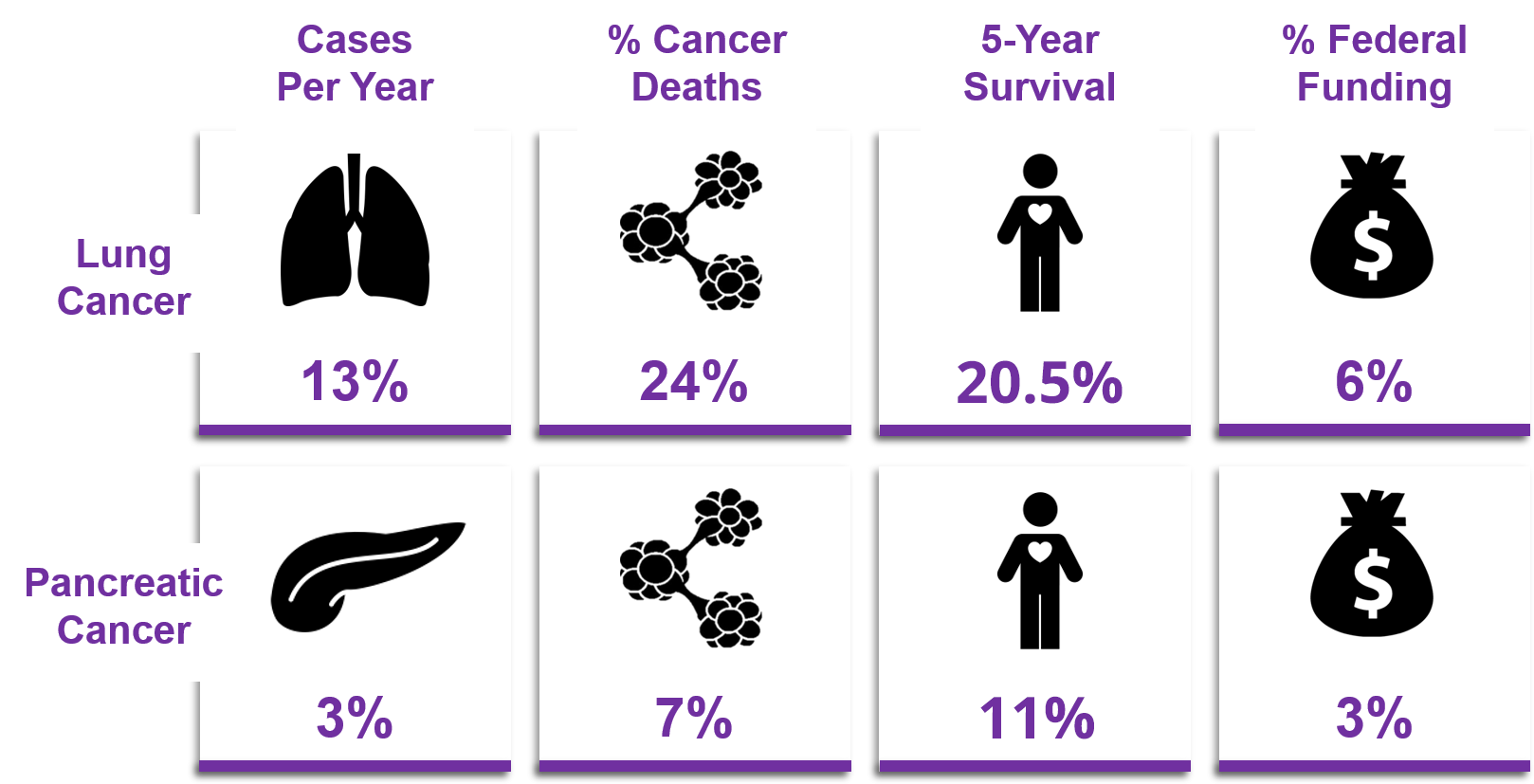
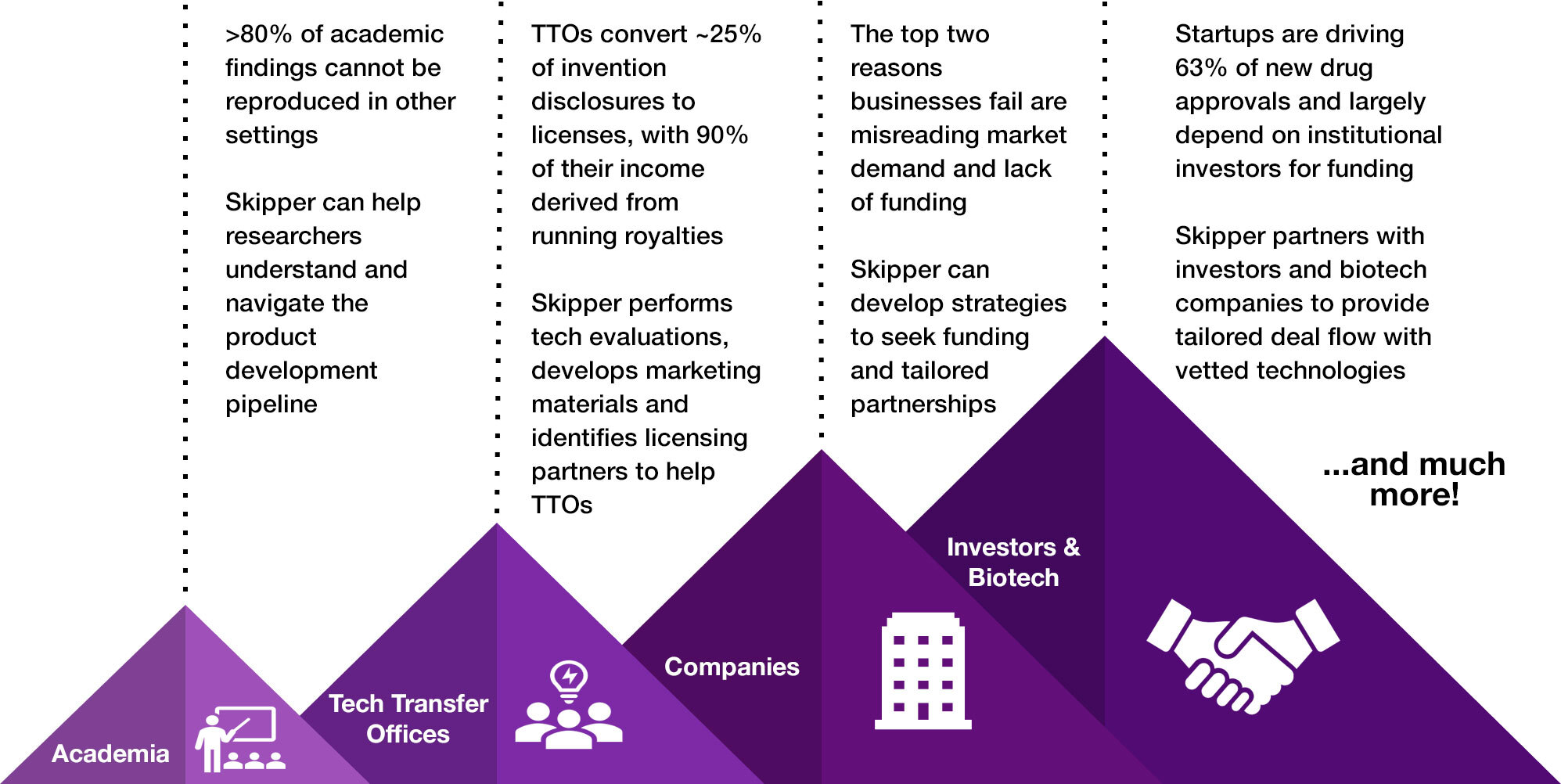
Oncology product development often takes the better part of a decade and upwards of hundreds of millions of dollars due to the scientific, financial, regulatory, safety, etc. barriers that must be navigated, and often not just once. Although it can be intimidating, Skipper Bio Med was founded on the premise that more research would successfully translate to patients and improve their outcomes if only there were more accessible resources to help navigate the journey. With our no-strings-attached, pro bono model, we are here to help you develop a personalized strategic roadmap, connect you with additional experts that you will need, and successfully overcome the challenges that have been holding you back.